
March 2, 2026
Process Plant Terminology Part 5: Mass Transfer Processes
This is the fifth of nine blog posts that provide layman’s descriptions of process engineering terminology commonly encountered when describing industrial processes and associated equipment. The terms described herein can be found in use across a wide range of processing and manufacturing industries, including oil and gas, refinery, petrochemical, chemical, mining and mineral, wastewater treatment, and power generation industries.
The first post provided an overview of the various drawing types that are used to depict industrial processing and manufacturing plants, while the second post discussed general processing and manufacturing plant terminology. Posts three through nine discuss common unit operations and processing equipment typically found in industrial processing and manufacturing plants, with this fifth post specifically discussing mass transfer processes.
Mass transfer processes involve the transfer of a chemical species of interest from one process stream to another, where the two streams are typically of different phases (i.e., mass transfer from a gas stream to a liquid stream, from a liquid stream to a gas stream, etc.). Chemical potential, or more practically chemical concentration, is the driving factor for mass transfer processes, with the chemical species moving from the higher-concentration stream (phase) to the lower-concentration stream (phase).
Mass transfer processes are typically employed to separate or purify chemical species of interest from a chemical mixture. Mass transfer processes commonly found in processing and manufacturing plants include distillation, absorption and stripping, adsorption and desorption, extraction, and membrane separation processes.
Industrial applications of mass transfer processes typically utilize industrial equipment such as a circular column to facilitate contact between the two (or more) phases of interest, with the columns designed such that they contain the equivalent of multiple stages of contact between the two phases in order to reach high levels of purity for the chemical species of interest. These multiple stages of contact are typically achieved using either multiple equilibrium stages (e.g., bubble caps as shown below in Figure 1) or various column packings (as shown below in Figure 2) to increase the surface area available for contact between the two phases. As shown below in Figure 2, column packing material can be either random (e.g., Raschig rings, Pall rings, or Dixon rings as shown below in Figure 2a–c, respectively) or structured packing, as shown below in Figure 2d.
Figure 1: Equilibrium Stages in a Mass Transfer Process1
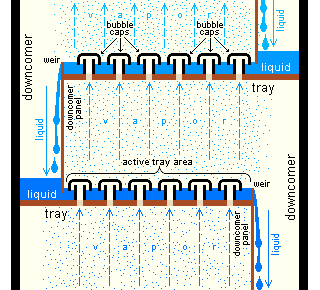
Figure 2: Typical Column Packing Materials2

Distillation
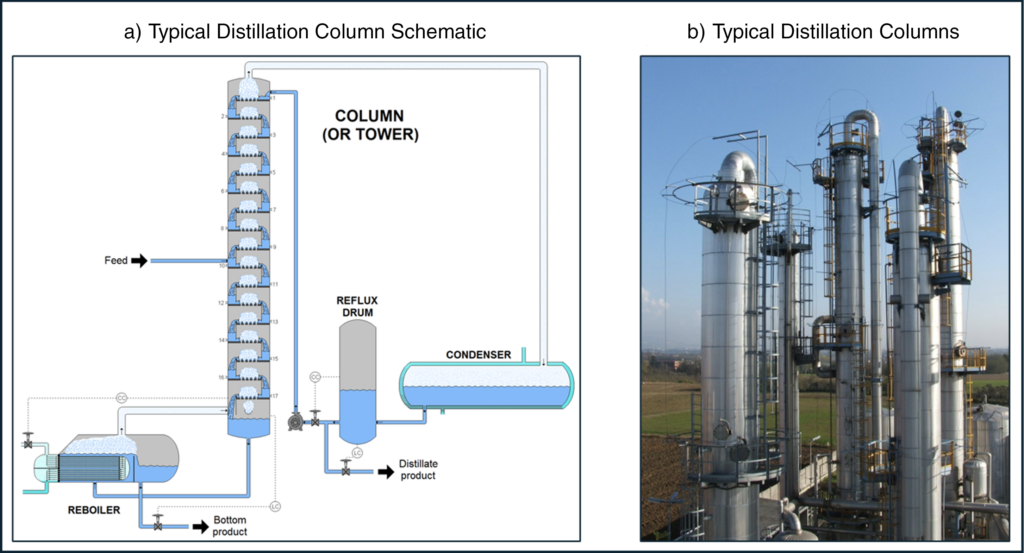
Distillation, which can be performed either in continuous mode or batch mode, is a separation process in which a feed mixture (typically either liquid phase or a mixture of liquid and vapor phases) is separated and purified based on the relative volatility of the chemical constituents in the mixture. Multi-stage continuous distillation, shown below in Figure 3, is also known as fractionation. Distillation processes are common in the oil refining, alcohol production, and chemical purification industries.
A typical schematic of a distillation column is shown below in Figure 3a. Heat is added to the column via a reboiler, typically using steam, to facilitate the vaporization (boiling) of the more volatile components of the feed mixture. Vaporized components rise upwards through the column while liquid components fall downward through the column, facilitating the separation. Multiple vapor/liquid contact points (trays) are utilized to increase the extent of separation. The distillate (vapor) product is typically condensed such that both the distillate (more volatile) and bottoms (less volatile) products are liquid phase. A portion of the distillate may be recycled (refluxed) back to the column to increase product purities. Higher reflux ratios (ratio of reflux flow rate to distillate product flow rate) improve product purity at the expense of increased energy requirements.
Figure 3: Typical Distillation Process3
Absorption and Stripping
Absorption is a process in which components of interest are transferred from one bulk material to another. In process engineering, absorption processes typically involve the mass transfer of gas phase components of interest to a liquid phase through either a physical or chemical mechanism. Absorption processes are typically utilized in pollution control applications (acid gas and/or volatile organic compound removal) and other purification applications, and are commonly found in the chemical processing and manufacturing, oil refining, and power generation industries.
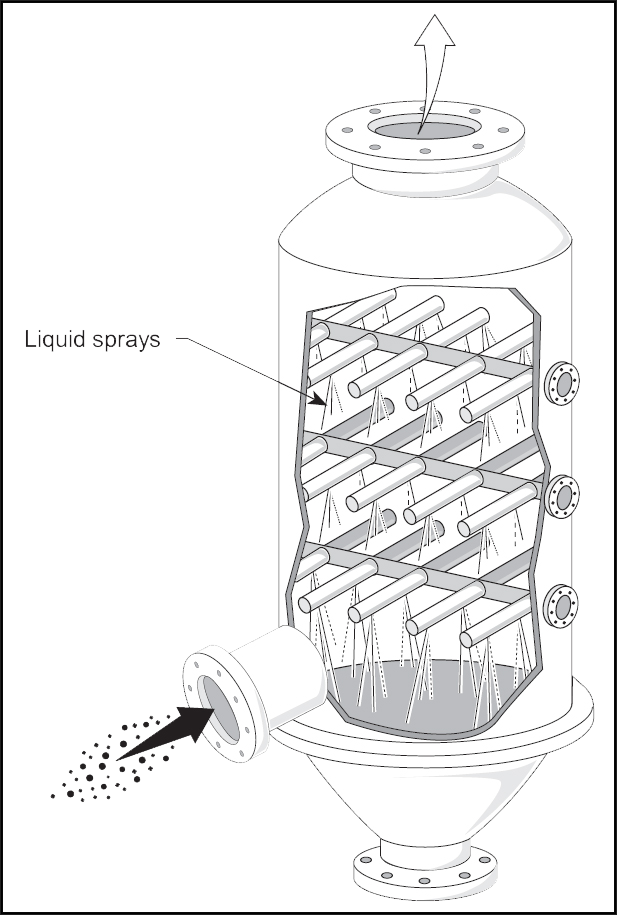
Absorption can be performed in a packed tower or, more commonly, in a spray tower, as shown below in Figure 4. In an absorber, the gas typically flows upwards and contacts downward-flowing liquid, with the chemical composition of the liquid (sometimes referred to as the sorbent or solvent) being selected such that it has an affinity for compounds of interest in the gas flow (e.g., the use of a caustic liquid such as sodium hydroxide to capture acid gases such as sulfur dioxide or carbon dioxide). The gaseous components of interest are transferred to (captured by) the liquid phase such that the gas leaving the absorber is relatively free of the components of interest.
Figure 4: Cross-Section of a Typical Counter-Flow Absorber Spray Tower4
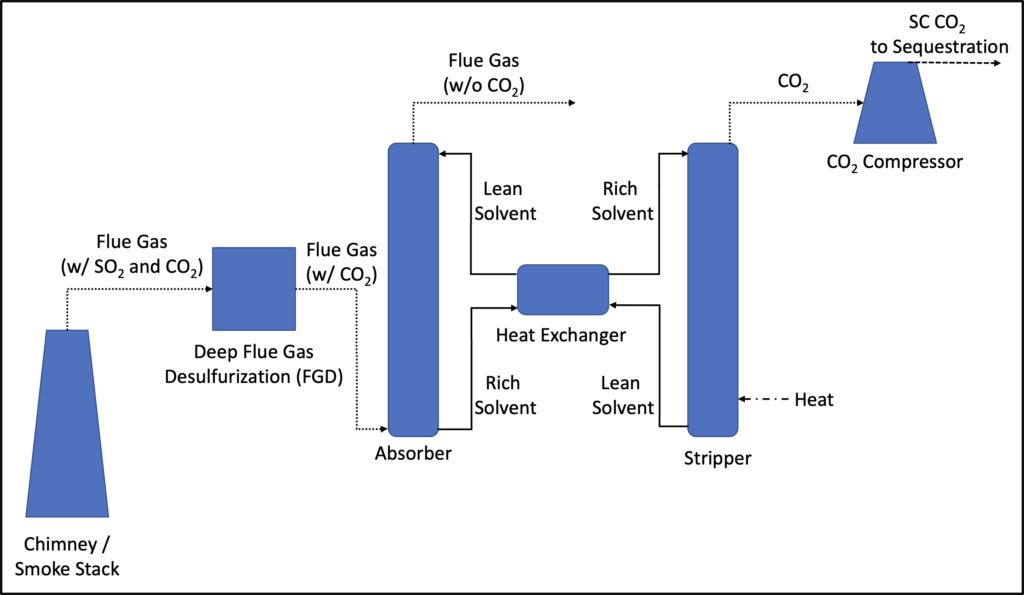
Stripping, the counterpart to absorption, is a process that involves the mass transfer of components of interest from a liquid stream to a vapor stream. Similar to absorption processes, stripping processes are typically carried out in packed towers or spray columns. Stripping processes are commonly found in wastewater treatment applications and are also sometimes coupled with absorption processes for solvent regeneration, as is the case for carbon capture applications, as shown below in Figure 5.
Figure 5: Typical Post-Combustion Carbon Capture System
Adsorption and Desorption
Adsorption is the adhesion of molecules to a surface. Adsorption differs from absorption because, whereas absorption involves mass transfer into a bulk fluid, adsorption involves mass transfer to a surface of interest. As shown below in Figure 6, common adsorbent materials include activated carbon, silica, and zeolite materials. High porosity and surface area are key characteristics of efficient adsorbents.
Adsorption processes are typically used in water purification (e.g., dissolved contaminant removal), air purification (e.g., volatile organic compound (VOC) and hydrogen sulfide (H2S) removal), and industrial drying applications (e.g., water removal) and are commonly found in the water treatment and chemical processing and manufacturing industries. Furthermore, chromatography, which is a type of adsorption process in which components of interest are adsorbed onto a column packing at various strengths based on the components’ affinity for the packing, is a separation and purification process commonly used in the pharmaceutical industry.
Figure 6: Common Adsorbents5

Desorption, the counterpart to adsorption, is typically employed to regenerate the adsorbent material to facilitate further adsorption. Desorption is typically accomplished either through heat addition, pressure reduction, or change in chemical characteristics such as pH.
Extraction
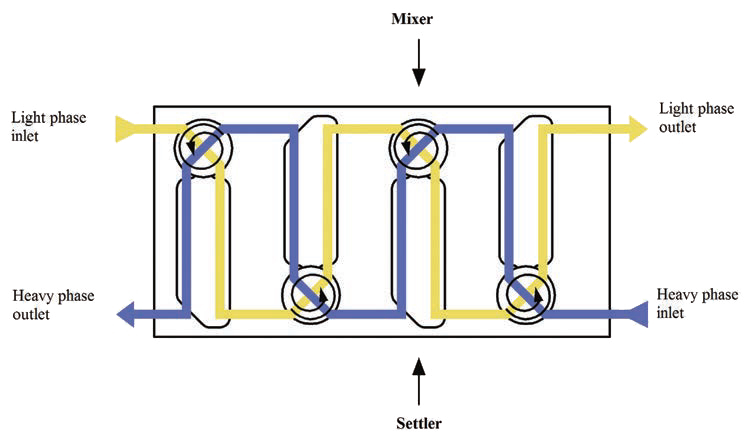
Liquid-liquid extraction (LLE) is a separation and purification process in which the mass transfer of components of interest occurs between two immiscible liquid phases based on the relative solubilities of the components in each liquid phase. LLE processes are common in the pharmaceutical, chemical manufacturing, mining, and water purification industries. Continuous LLE processes typically employ a train of mixer-settlers, where the immiscible liquids are first mixed and then allowed to settle back to two distinct phases to facilitate multiple stages of mass transfer, as shown below in Figure 7.
Figure 7: Typical LLE Mixer-Settler Configuration6
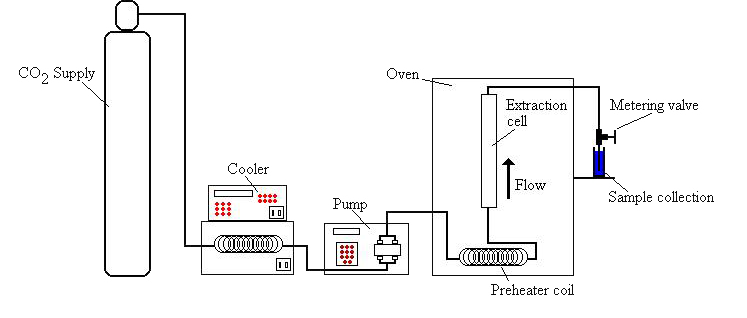
Supercritical fluid extraction (SFE) is an extraction process that utilizes a supercritical fluid to extract components of interest from a solid or liquid material. Supercritical fluids are defined as substances where both the temperature and pressure of the fluid exceed the substances’ critical temperature and pressure, respectively. Supercritical fluids typically exhibit high, liquid-like densities while exhibiting gas-like diffusion properties, making them powerful solvents. Industrial applications of SCF extraction include the use of supercritical carbon dioxide (SCO2) in dry cleaning and coffee decaffeination applications. A typical schematic of an SCO2 extraction system is shown below in Figure 8.
Figure 8: Typical SCO2 Extraction System7
Membrane Separations
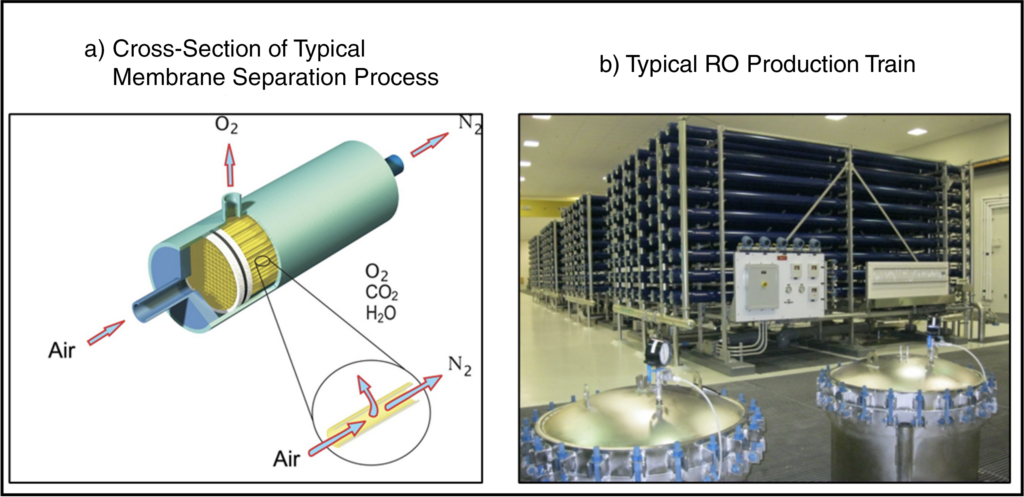
Membrane separation processes employ semi-permeable barriers (membranes) that selectively allow the mass transfer of components of interest through the membrane. Membrane separations are typically driven by gradients in pressure, concentration, or electric potential. Because membrane separation processes typically do not require heat, they are less energy-intensive than other separation processes such as distillation. Reverse osmosis (RO), which applies pressure across the membrane to remove contaminants, is an important membrane separation process in the water purification industry. Membrane separation processes are also common in the biotechnology and pharmaceutical industries.8 A cross-section of a typical membrane separation process and a typical RO production train are shown below in Figure 9a and Figure 9b, respectively.
Figure 9: Typical Membrane Separation Systems9
1 “Typical bubble cap trays used in industrial distillation columns” by H Padleckas under CC BY-SA 2.5, accessed February 12, 2026.
2 Adapted from “Random column packing” and “Structured packing,” Wikipedia: a) “This shows a photo of Raschig rings” by Chemical Engineer under Public Domain, b) “Pall rings (beige and large white) and Bialecki rings (others)” by LukaszKatlewa under CC BY-SA 3.0, c) “Dixon rings” by Joeravo under CC BY-SA 4.0, d) “Structured packing” by Luigi Chiesa under CC BY-SA 3.0. Accessed February 12, 2026.
3 Adapted from “Fractionating Column,” Wikipedia: a) “Figure 6: Entire view of a Distillation Column” by MAG, Ch.E. under CC BY-SA 3.0, b) “Figure 2: Typical industrial fractionating columns” by Luigi Chiesa under CC BY 3.0. Accessed February 12, 2026.
4 “Typical countercurrent-flow spray tower.” by United States Environmental Protection Agency (US EPA) under Public Domain. Accessed February 12, 2026.
5 Adapted from “Adsorption,” “Silica Gel,” and “Zeolite,” Wikipedia: a) “Activated carbon is used as an adsorbent” by Ravedave under CC BY-SA 2.5, b) “Silica gel beads sparkling in light” by Wiebew under CC BY-SA 3.0, and c) “Synthetic zeolite” by Seaterror under CC BY-SA 3.0. Accessed February 12, 2026.
6 “Four-stage battery of mixer-settlers for counter-current extraction” by Perdula under CC BY-SA 3.0. Accessed February 12, 2026.
7 “Figure 1. Schematic diagram of SFE apparatus” by Stainless316 under Public Domain. Accessed February 12, 2026.
8 See “Membrane Technology,” Wikipedia. Accessed February 16, 2026.
9 Adapted from “Membrane Technology” and “Reverse Osmosis,” Wikipedia: a) “Separation of air into oxygen and nitrogen through a membrane” by GRASYS under CC BY-SA 3.0, b) “RO production train, North Cape Coral Reverse Osmosis Plant” by Twhair under CC BY-SA 4.0.
ADDITIONAL RESOURCES

Blog
Discover industry insights on construction disputes and claims, project management, risk analysis, and more.
MORE

Articles
Articles by our engineering and construction claims experts cover topics ranging from acceleration to why claims occur.
MORE

Publications
We are committed to sharing industry knowledge through publication of our books and presentations.
MORE
RECOMMENDED READS
Project and Construction Management Terminology
This post is part of a series defining industry terminology. This post addresses project and construction management.
READ
A Tale of Two Claims: Same Contract, Two Fates
The success of a claim often hinges not on what happened in the field, but how it was documented, analyzed, and communicated.
READ
The Role and Benefit of a Consultant in a Construction Project: Part 1
This is the first blog post in a two-part series on the role and benefit of a construction consultant.
READ

